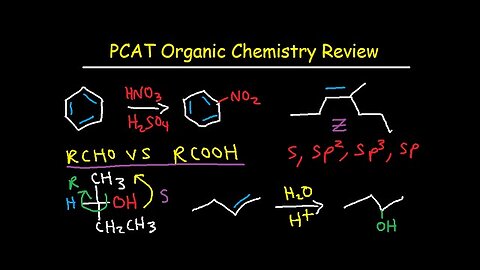
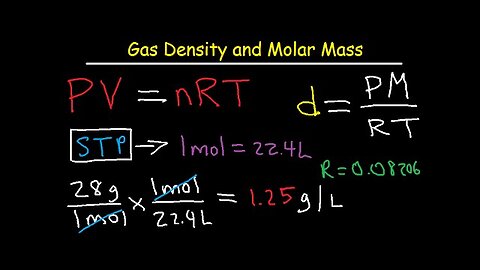PCAT Organic Chemistry Review Study Guide
This video tutorial focuses on the organic chemistry section of the PCAT. It provides a nice review of the first two semesters of organic chemistry.
Here is a list of topics:
1. Nomenclature and Functional Groups
2. Hybridization, Bond Angle, and Molecular Geometry
3. Sigma and Pi Bonds, Bond Strength vs Length
4. Acids and Bases Periodic Trend - pKa
5. Resonance Structures - Curve Arrow Notation
6. Newman Projections of Butane - Gauche, Anti, Staggered, Eclipse
7. Conformations of Cyclohexane - Chair, Half Chair, Twist Boat
8. Torsional vs Steric Strain and 1,3 Diaxial Interactions
9. Potential Energy Diagrams - Kinetics and Rate Law
10. Catalyst, Activation Energy, Rate Determining Step
11. Initiation, Propagation, and Termination
12. Bromination vs Chlorination Selective vs Nonselective Reactions
13. Stereochemistry - Enantiomers, Diastereomers, and Meso Compounds - R / S Configuration, Chirality and Optical Activity
14. Electrophilic Addition Reactions of Alkenes and Alkynes
15. Acid Catalyzed Hydration, Hydroboration Oxidation, Oxymercuration Demercuration, and Ozonolysis
16. SN2, SN1, E1, and E2 reaction mechanism
17. Polar protic vs aprotic solvents - Nucleophiles and Leaving Groups
18. Inversion of Configuration vs Racemic Mixture
19. Oxidation and Reduction Reaction of Alcohols
20. Grignard & Gilman Reagents, DIBAH and Organolithium reagents
21. conjugate addition - 1,3 butadiene - Kinetic vs Thermodynamic Product
22. Diels Alder Reaction - Examples and Practice Problems
23. Acid Catalyzed Cleavage of Ethers and Epoxides
24. Nucleophilic Addition of Carbonyl Compounds - Aldehydes and Ketones
25. Enol and Enolate Reactions - Alpha Hydrogen Substitution
26. Wittig Reaction, HVZ Reaction, Haloform Reaction
27. Iodoform Test - Methyl Ketones
28. Tollen's Reagent Test for Aldehydes
29. Imine vs Enamine - Primary and Secondary Amines
30. Gabriel Synthesis Reaction
31. Hoffman vs Curtius Rearrangement Reactions
32. Carboxylic Acid Derivatives Review
33. Benzene Reactions - Electrophilic and Nucleophilic Aromatic Substitution Reactions
34. Nitration, Sulfonation, Chlorination, Bromination, Friedel Crafts Alkylation and Acylation Reactions
35. Side Chain Reactions of Benzene
36. Benzyne Intermediate vs Meisenheimer Complex
37. Aldol Condensation Reaction
38. Claisen Ester Reaction
39. Acetoacetic and Malonic ester Reaction Mechanism
40. Michael Reaction
41. Robinson Annulation Reaction Mechanism
60
views
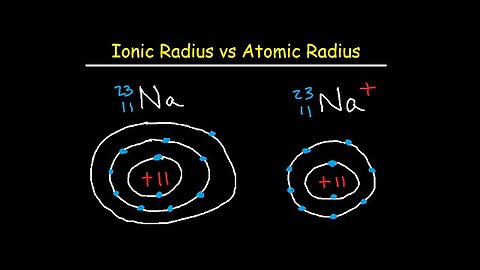
Ionic and Atomic Radius - Periodic Trends
This chemistry video tutorial focuses on periodic trends such as ionic radius and atomic radius. It provides plenty of examples and explains the fundamental concepts of why some ions are smaller than atoms while others are larger. Here is a list of topics
1. Atomic Radii decreases left to right
2. Effective Nuclear Charge, Atomic Number, and Number of Protons
3. Atomic Radius Increases from top to bottom due to additional energy levels
4. Neutral vs Positively Charged Ion / Cation – Number of Shells or Energy Levels
5. Neutral Parent Atom vs Negatively Charged Ion – Electron Cloud Expansion
6. General Ionic Radii Trend – Cations are smaller than Anions
Chemistry - Basic Introduction:
• Chemistry
Stoichiometry Practice Problems:
• How To Solve Stoichiometry Problems
Molarity Practice Problems:
• Molarity Practice Problems
Ideal Gas Law Problems:
• Ideal Gas Law Practice Problems
Basic Calorimetry Problems:
• How To Solve Basic Calorimetry Proble...
Enthalpy of Formation & Heat Combustion:
• Enthalpy of Formation Reaction & Heat...
Speed of Light, Frequency, & Wavelength:
• Speed of Light, Frequency, and Wavele...
Intro to Electron Configuration:
• Electron Configuration - Basic introd...
Quantum Numbers - Practice Test:
• Orbitals, Quantum Numbers & Electron ...
_____________________________________________________________________________________________
Ionization Energy:
• Ionization Energy - Basic Introduction
Electron Affinity:
• Electron Affinity Trend, Basic Introd...
Atomic Radius:
• Atomic Radius - Basic Introduction - ...
Bond Energy & Bond Length:
• Bond Energy & Bond Length, Forces of ...
Electronegativity:
• Electronegativity, Basic Introduction...
Periodic Trends:
• Ionization Energy Electron Affinity A...
Polar & Nonpolar Covalent Bonding:
• Polar Covalent Bonds and Nonpolar Cov...
Bond Polarity & Dipole Moment:
• Bond Polarity, Electronegativity and ...
Ionic Radius:
• Ionic Radius Trends, Basic Introducti...
Lattice Energy:
• Lattice Energy of Ionic Compounds, Ba...
Born Haber Cycle:
• Born Haber Cycle, Basic Introduction,...
Bond Energy Calculations:
• Bond Energy Calculations & Enthalpy C...
How To Draw Lewis Structures:
• How To Draw Lewis Structures
VSEPR Theory:
• VSEPR Theory - Basic Introduction
Molecular Geometry:
• Molecular Geometry & VSEPR Theory - B...
Lewis Dot Structures:
• Lewis Dot Structures - How To Calcula...
Lewis Structures of Ionic Compounds:
• How To Draw The Lewis Structures of I...
Octet Rule Exceptions:
• Exceptions To The Octet Rule - Lewis ...
Resonance Structures:
• Resonance Structures, Basic Introduct...
Polar and Nonpolar Molecules:
• Polar and Nonpolar Molecules
Formal Charge Calculations:
• How To Calculate The Formal Charge of...
Lewis Structures - Mega Review:
• Lewis Structures, Introduction, Forma...
_____________________________________________________________________________________________
Hybridization of Atomic Orbitals:
• Hybridization of Atomic Orbitals - Si...
Molecular Orbital Theory:
• Molecular Orbital Theory - Bonding & ...
Dipole Dipole Forces of Attraction:
• Dipole Dipole Forces of Attraction - ...
Hydrogen Bonding:
• Hydrogen Bonds In Water Explained - I...
Unit Cell Chemistry:
• Unit Cell Chemistry Simple Cubic, B...
General Chemistry 1 Final Exam Review:
• General Chemistry 1 Review Study Guid...
67
views
Net Ionic Equation Worksheet and Answers
This chemistry video tutorial focuses on how to write net ionic equations. It provides a worksheet full of examples and practice problems along with the answers to the questions and how to get them.
Useful Textbooks: https://www.amazon.com/shop/theorgani...
E-Book & E-mail Newsletter: https://www.video-tutor.net/math-and-...
Subscribe To My YouTube Channel: https://bit.ly/37WGgXl
_______________________________________________________________________________________________
Here is a list of topics:
1. Net Ionic Equation of Double Replacement Reactions
2. How to Balance the Formula of an Ionic Compound
3. How to predict the products of a double displacement reaction
4. How to write the total ionic equation for a precipitation reaction
5. How to identify and remove the spectator ions
6. How to write the net ionic equation for an acid base reaction
7. Predicting the products and balancing the neutralization reaction
8. Net Ionic Equations – Strong acids and weak acids – low ionization / dissociation
9. Solubility Rules for net ionic equations
10. Writing Net Ionic Equations for Single Replacement Reactions – Redox
11. Diatomic Halogen Redox Reactions
11. Gas forming reactions – net and total ionic equations – CO2 and NH3
________________________________________________________________________________________________
Chemistry - Basic Introduction:
• Chemistry
Protons, Neutrons, & Electrons:
• How To Calculate The Number of Proton...
Writing Formulas - Ionic Compounds:
• Writing Chemical Formulas For Ionic C...
Balancing Chemical Equations:
• How To Balance Chemical Equations
Basic Stoichiometry:
• Stoichiometry Basic Introduction, Mol...
Stoichiometry Practice Problems:
• How To Solve Stoichiometry Problems
Solute, Solvent, & Solution:
• Solute, Solvent, & Solution - Solubi...
Strong & Weak Electrolytes:
• Identifying Strong Electrolytes, Weak...
Molarity Practice Problems:
• Molarity Practice Problems
Ion Concentration In Solutions:
• Ion Concentration in Solutions From M...
Dilution Problems:
• Dilution Problems, Chemistry, Molarit...
Types of Chemical Reactions:
• Types of Chemical Reactions
Solubility Rules:
• Solubility Rules
Predicting The Products of Reactions:
• Predicting The Products of Chemical R...
Activity Series of Metals:
• Activity Series of Metals - Chemistry
Will This Reaction Occur?
• Chemistry - Will The Reaction Occur?
Predicting Products of SR Reactions:
• Predicting Products of Single Replace...
Double Replacement Reactions:
• Introduction to Double Replacement Re...
Net Ionic Equations:
• Precipitation Reactions & Net Ionic E...
Writing Chemical Equations From Words:
• How To Write Chemical Equations From ...
Na2CO3 + HCl:
• Na2CO3 + HCl - Sodium Carbonate + Hyd...
Solution Stoichiometry:
• Solution Stoichiometry - Finding Mol...
Molarity & Dilution Problems:
• Molarity Dilution Problems Solution S...
Acid Base Neutralization Reactions:
• Acid Base Neutralization Reactions & ...
Acid Base Titration Problems:
• Acid Base Titration Problems, Basic I...
Mixture Problems:
• Mixture Problems
Calculating Oxidation Numbers:
• How To Calculate Oxidation Numbers - ...
Oxidation and Reduction Reactions:
• Oxidation and Reduction Reactions - B...
Balancing Redox Reactions:
• Half Reaction Method, Balancing Redox...
Ideal Gas Law Problems:
• Ideal Gas Law Practice Problems
General Chemistry 1 Final Exam Review:
• General Chemistry 1 Review Study Guid...
Affiliate Marketing Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
64
views
Gas Laws - Equations and Formulas
This video tutorial focuses on the equations and formula sheet that you need for the gas law section of chemistry. It contains a list of equations as well as the fundamental concepts, relationship, and units that you need to know as well.
Useful Textbooks: https://www.amazon.com/shop/theorgani...
E-Book & E-mail Newsletter: https://www.video-tutor.net/math-and-...
Subscribe To My YouTube Channel: https://bit.ly/37WGgXl
_______________________________________________________________________________________________
Here is a list of topics:
1. Pressure, Force, and Area
2. Units of Pressure – atm, torr, mmhg, kpa, and pa
3. Ideal Gas Law Formula - PV=nRT
4. Gas Law Constant R=0.08206 or 8.3145 – Units of R
5. Combined Gas Law Equation – Pressure, Volume, Moles, and Temperature
6. Boyle’s Law Equation – Inverse Relationship between Pressure and Volume
7. Charles Law Formula – Direct Proportion – Temperature and Volume
8. Gay Lussac Law Equation – Pressure and Temperature – Direct Linear Relationship
9. Avogadro’s Law – Moles and Pressure
10. The relationship between pressure and number of collisions
11. Dalton’s Law of Partial Pressure and Total Pressure
12. Mole Fraction of a gas and Partial Pressure
13. Gas Density Formula and Molar Mass – g/L
14. Molar mass of a gas given grams
15. Density of Gas versus Pressure, Molar Mass, and Kelvin Temperature
16. STP – Standard Temperature and Pressure
17. Molar Volume – 1 mole of gas equals 22.4 Liters
18. Average Kinetic Energy of a Gas Formula and Temperature
19. The Root Mean Square Velocity Equation / Formula
20. Speed, Temperature, and Molar Mass Relationship
21. Rate of Effusion vs Gas Diffusion
22. Graham’s Law of Effusion – Rate vs Molar Mass – Units of M – Kg/mol
________________________________________________________________________________________________
Chemistry - Basic Introduction:
• Chemistry
Stoichiometry Practice Problems:
• How To Solve Stoichiometry Problems
Molarity Practice Problems:
• Molarity Practice Problems
______________________________________________________________________________________________
Pressure & Boiling Point:
• Introduction to Pressure - Force & Ar...
Gas Pressure Unit Conversion:
• Gas Pressure Unit Conversions - torr ...
Boyle's Law Practice Problems:
• Boyle's Law Practice Problems
Charles Law:
• Charles' Law
Gay Lussac's Law:
• Gay Lussac's Law Practice Problems
Avogadro's Law:
• Avogadro's law Practice Problems
Ideal Gas Law Problems:
• Ideal Gas Law Practice Problems
Combined Gas Law Problems:
• Combined Gas Law Problems
Gas Stoichiometry Problems:
• Gas Stoichiometry Problems
Molar Mass of a Gas at STP:
• Molar Mass of a Gas at STP - Equation...
Gas Density at STP:
• Gas Density and Molar Mass Problems a...
Dalton's Law of Partial Pressure:
• Dalton's Law of Partial Pressure Prob...
Collecting Gas Over Water:
• Collecting Gas Over Water Practice Pr...
Gas Density of Mixtures:
• Gas Density & Average Molar Mass of a...
Average Kinetic Energy of a Gas:
• Average Kinetic Energy of a Gas and R...
Graham's Law of Effusion:
• Graham's Law of Effusion
Kinetic Molecular Theory of Gases:
• Kinetic Molecular Theory of Gases - P...
Gas Law Problems Review:
• Gas Law Problems Combined & Ideal - D...
___________________________________________________________________________________________
Basic Calorimetry Problems:
• How To Solve Basic Calorimetry Proble...
Enthalpy Problems:
• Enthalpy Change of Reaction & Formati...
General Chemistry 1 Final Exam Review:
• General Chemistry 1 Review Study Guid...
____________________________________________________________________________________________
Affiliate Marketing Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
79
views
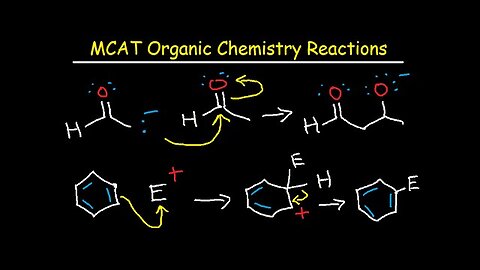
MCAT General Chemistry Lectures Review Prep Part 2 – Equations & Practice Questions
This mcat review prep video tutorial focuses on the general chemistry section of the mcat. This is the part 2 version and it contains plenty of notes, concepts, equations, formulas, example problems and practice questions. Here is a list of topics included in this video:
Full Video: https://vimeo.com/ondemand/mcatcourse...
1. State Functions – Enthalpy, Entropy, and Free Energy
2. Internal Energy of a System – Heat Transfer and Work
3. System vs Surroundings – Endothermic and Exothermic Processes
4. Pressure, Volume and Work – Gas Expansion vs Compression
5. Temperature Change vs Phase Change – Heat Transfer Problems
6. Heat of Vaporization and Enthalpy of Fusion
7. Freezing, Melting, Vaporization, Condensation, Sublimation and Deposition
8. Heats of Formation, Enthalpy of Reaction, Hess Law, Calorimetry, and Bond Dissociation Energy
9. Quantum Mechanics – n, l, ml, and ms. S, P, D, F orbitals. Energy Levels and Sublevels.
10. Ground State Electron Configuration vs Noble Gas Notation With Orbital Diagrams
12. Radio Waves, Microwaves, Infrared, Visible Light, Ultraviolet, X-Rays, and Gamma Rays
13. Electromagnetic Radiation – Frequency, Wavelength, Energy of a Photon, and Speed of Light
14. Ionization Energy, Electron Affinity, Atomic Size, Ionic Radii, Metallic Character, & Electronegativity
15. Lewis Structures, Hybridization, Molecular Geometry, Bond Angle, Polar and Nonpolar
16. Formal Charge Calculations, Bond Order, Sigma and Pi Bonds, Bond Length vs Strength
17. Intermolecular Forces – Ion Dipole, Hydrogen Bonds, Dipole Dipole and London Dispersion Forces
18. Boiling Point vs Vapor Pressure – Molar Mass, Polarizability, and Temporary Induced Dipoles
19. Classius Clayperon Equation
20. Vapor Pressure and Temperature, Boiling Point vs Elevation
21. Molality, Molarity, Mass %, and Density Calculations – Solute, Solvent, and Solutions
22. Henry’s Law – Gas Solubility vs Pressure vs Temperature
23. Colligative Properties – Boiling Point Elevation, Freezing Point Depression
24. Vapor Pressure Lowering – Raoult’s Law and Osmotic Pressure
25. Phase Diagrams – Critical Point, Triple Point, Normal Melting Point, and Normal Boiling Point
37
views
PCAT General Chemistry Review Test Prep Study Guide Course
This study guide tutorial focuses on the general chemistry section of the PCAT – Pharmacy College Admission Test. This review contains plenty of example problems and practice questions along with notes, formulas & equations, plus the fundamental chemistry concepts to go with it.
Here is a list of topics included in this video:
1. Atoms, Molecules, and Ionic Compounds
2. Ionic Bonding, Polar and Nonpolar Covalent Bonding and Coordinate Covalent Bonds
3. Pure Substances vs Mixtures – Heterogeneous and Homogeneous
4. Allotropes, Alloys, and Isotopes
5. Alkali Metals, Alkaline Earth Metals, Transition Metals, Halogens, Chalcogens, and Noble Gases
6. Periodic Table – Metals, Metalloids, and Nonmetals
7. Celsius, Kelvin, and Fahrenheit Temperature Conversions
8. Unit Conversion, Dimensional Analysis, and the Metric System
9. Density Practice Problems
10. Mole to Mole and Gram to Gram Stoichiometry
11. Molar Mass, Atomic Mass, Molecular Weight and Formula Weight
12. Average Atomic Mass of Isotopes and Relative Percent Abundance
13. Empirical Formula, Molecular Formula, Mass Percent, and % Composition
14. Limiting and Excess Reactant Stoichiometry Problems
15. Synthesis and Decomposition Reactions
16. Single Replacement and Double Displacement Reactions
17. Oxidation Numbers of Elements in Compounds and Polyatomic Ions
18. Oxidizing Agents and Reducing Agents – Redox Reactions
19. Molarity Concentration Problems, Acid Base Titrations, and Dilution
20. Solution Stoichiometry, Net Ionic Equations, and Spectator Ions
21. Precipitation Reactions, Acid Base Neutralization Reactions and Gas Evolution Reactions
22. Strong, Weak, and Nonelectrolytes
23. Ideal Gas Law and Combined Gas Law
24. Dalton’s Law of Partial Pressures and Graham’s Law of Effusion
25. Average Kinetic Energy of Gases and Temperature
26. Relationship Between Temperature, Pressure, Volume and Moles
27. Boyle’s Law, Charles Law, Gay Lussac Law, and Avogadro’s Gas Laws
28. Root Mean Square Velocity Calculations
29. Density of Gas at STP and Molar Mass Calculations
30. Kinetic Molecular Theory of Ideal Gases.
31
views
DAT General Chemistry Review
This online course video tutorial review focuses on the general chemistry section of the DAT Exam – the Dental Admission Test. It provides plenty of notes along with the fundamental concepts of topics such as matter, stoichiometry, chemical reactions, and gas law chemistry. It contains a list of equations, formula sheets, along with explanations on how to use the formulas the right way. This study guide contains plenty of examples and practice questions and hard problems for you to do well on the DAT exam.
Full DAT Course Review:
https://vimeo.com/ondemand/datreviewc...
Here is a list of topics:
1. Atoms, Molecules and Ionic vs Molecular Compounds
2. Pure Substances vs Heterogeneous and Homogeneous Mixtures
3. Ionic vs Covalent Bonding plus Coordinate Covalent Bonds
4. Isotopes, Allotropes, Alloys, Intensive and Extensive Properties
5. Characteristics of Metals, Metalloids, and Nonmetals
6. Periodic Table – Alkali Metals, Alkaline Earth, Chalcogens, Halogens, and Noble gases
7. Density Practice Problems, Unit Conversion, Dimensional Analysis and the Metric System
8. Temperature Conversions, Celsius, Kelvin and Fahrenheit
9. Grams, Moles, Atoms, Particles, Ions, Molecules, and Formula Unit Conversions
10. Mole to Mole and Gram to Gram Stoichiometry
11. Molar Mass, Atomic Mass, Molecular Weight and Formula Weight Calculations
12. Average Atomic Mass and Relative Percent Abundance of Isotopes
13. Phases of Matter – Solids, Liquids, Gases, and Plasma
14. Actual Yield, Theoretical Yield, and Percent Yield Calculations
15. Mass %, Percent Composition, and Empirical Formula Determination – Combustion Analysis
16. Limiting and Excess Reactant Stoichiometry Problems
17. Synthesis / Combination and Decomposition Reactions
18. Oxidation Numbers and States for Elements, Compounds, and Polyatomic Ions
19. Nomenclature of Ionic Compounds, Molecular Compounds, and Acids
20. Polar and Nonpolar Covalent Bonds
21. Redox Reactions – Oxidation vs Reduction – How to Identify The Oxidizing and Reducing Agent
22. Single Replacement and Double Displacement Reactions Including Combustion Reactions
23. Precipitation, Acid-Base Neutralization, and gas evolution reactions
24. Accuracy vs Precision & the activity series of metals
25. Molarity Calculations, Acid Base Titrations, and Dilution Problems
26. Solution Stoichiometry with Limiting and Excess Reactant
27. Net Ionic Equations and Spectator Ions
28. Strong, Weak, and Nonelectrolytes
29. Solubility Rules – Soluble vs Insoluble Compounds
30. How to Write the Formula for Ionic and Covalent Compounds Including Acids
31. Strong Acids, Weak Acids, Strong Bases, and Weak Bases
32. Acidic, Basic, and Neutral Salts
33. Arrhenius, Bronsted Lowry and Lewis Acids and Bases
34. Gas Laws – Ideal Gas Equation and the Combined Gas law formula
35. Boyle’s, Charles, Gay Lussac, and Avogradro’s Law Equations
36. Mole Fraction, Partial and Total Pressure
37. Dalton’s Law of Partial Pressures and Graham’s Law of Effusion
38. Gas Density at STP and Molar Mass Problems
39. How To Identify The Unknown Gas
40. Gas Stoichiometry Problems with Liter to Liter Conversions
41. Average Kinetic Energy of a Gas and Temperature
42. Root Mean Square Velocity of a Gas
43. Kinetic Molecular Theory of Ideal Gas
44. Temperature and Pressure Conditions For Ideal Gases
32
views
MCAT Organic Chemistry Review Reactions Summary Study Guide Part 2
This MCAT organic chemistry reactions review study guide tutorial provides plenty of examples, questions, and practice problems on reactions and their mechanisms.
Full Video: https://vimeo.com/ondemand/mcatcourse...
Here is a list of topics:
1. 1,2 vs 1,4 addition of HBr to 1,3-butadiene – Thermodynamic vs Kinetic Product
2. Diels Alder Reaction – 4 + 2 Cycloadditon Reaction
3. Benzene Reactions – Electrophilic and Nucleophilic Aromatic Substitution Reactions
4. Addition-Elimination vs Elimination-Addition Mechanism
5. Nitration, Sulfonation, Bromination, Chlorination, Iodination Reactions
6. Friedel Crafts Alkylation and Acylation Reaction Mechanisms
7. Diazotization of Aniline and AreneDiazonium Salts – Sandmeyer Reaction
8. Benzyne Intermediate vs Meisenheimer Complex
9. Ortho Para Directors vs Meta Directors
10. Reactions of Aldehydes and Ketones
11. Ozonolysis Reaction
12. Oxidation of Alcohols into Ketones, Aldehydes and Carboxylic Acids
13. Nitrile Reduction to Ketone Using a Grignard reagent
14. Imine vs Enamine – Primary and Secondary Amines with Ketones and Schiff Bases
15. Tollen’s Reagent Test – Aldehyde Selectivity
16. 2,4-DNP Test for Carbonyl Functional Groups
17. The Wittig Reaction
18. Carboxylic Acid Derivative Reactions – Acid Chlorides, Anhydrides, Amides, and Esters
19. Transesterification Reaction and the Acid Base Hydrolysis of Esters and Amides
20. Lactones and Lactams Ring Opening Reactions
21. Direct Alkylation of Ketones Using LDA and NaH with CH3Br – Thermodynamic vs Kinetic Enolate Ion
22. Alpha Hydrogen Substitution Reactions
23. Alpha Halogenation Reaction Mechanisms with Br2, Cl2 and I2 Using Hydroxide Base Catalyst
24. Haloform Reaction and the HVZ Hell Volhard Zelinski Reaction
25. The Stork Reaction – Direct Alkylation of Ketones Using Enamines
26. Iodoform Test for Methyl Ketones – Yellow Precipitate
27. Aldol Condensation Reaction
28. Mixed Aldol Reaction and Retro-aldol mechanism – base catalyzed
29. Canizzaro Reaction Mechanism
30. Claisen Ester Condensation Reaction – Formation of Beta Keto Esters
31. Acetoacetic Ester Synthesis Reaction to Produce Substituted Ketones
32. Decarboxylation of Beta Keto carboxylic Acids
33. Malonic Ester Synthesis Reaction – Formation of Substituted Carboxylic Acid Compounds
34. Robinson Annulation Reaction
35. Michael Reaction – Donor vs Acceptor
36. Cyclic Intramolecular Aldol Reaction Mechanism
37. Dieckmann Condensation Reaction Followed By Acid Hydrolysis and Decarboxylation to Produce Cyclohexanone
105
views
MCAT Test Prep General Chemistry Review Study Guide Part 1
This online video course tutorial focuses on the general chemistry section of the mcat. This video provides a lecture filled with notes and a sheet of formula and equations that will be useful as well as the fundamental concepts that go with. This study guide / review will help you to learn the most important topics in chemistry that you need to do well the physical science part of the MCAT. It has plenty of example problems and practice questions for you to test your knowledge on.
Full Video:
https://vimeo.com/ondemand/mcatcourse...
Here is a list of topics:
1. Atoms, Molecules, Pure Elements, Ionic and Covalent Compounds
2. Subatomic Particles – Electrons, Protons, and Neutrons
3. The difference between an atom and an ion
4. Cations vs Anions – Positive vs Negative Charged Ions
5. Pure Substance and Mixtures – Homogeneous and Heterogeneous
6. Density Practice Problems – Unit Conversion and Dimensional Analysis
7. Temperature Conversions – Celsius, Fahrenheit, and Kelvin
8. Atomic Number, Mass Number, and Charge
9. Isotopes – C12 and C13 – Hydrogen, Deuterium and Tritium
10. Allotropes of Carbon – Diamond and Graphite
11. Alloys of Metals – Bronze, Brass, Steel, and Nichrome Wire
12. 7 Diatomic Elements – H2, N2, O2, F2, Cl2, Br2, and I2
13. Monoatomic vs Polyatomic Ions – List to Memorize
14. Nomenclature of Ionic and Molecular Compounds
15. How To Write The Formula of Ionic and Covalent Compounds
16. Strong and Weak Acids vs Strong and Weak Bases
17. The pH Scale – Acidic vs Basic Conditions
18. How To Name Acids – Nomenclature & How To Write the Formula of an Acid
19. Acid Base Conjugate Pairs – Conjugate Acid vs Conjugate Base
20. Arrhenius Acid vs Arrhenius Base – H3O+ Hydronium Ions vs Hydroxide OH- Ions
21. Bronsted Lowry Acid vs Bronsted Lowry Base – Proton Donors vs Proton Accetors
22. Lewis Acid vs Lewis Base – Electron Pair Acceptors and Electron Pair Donors
23. Acid water reaction vs base water reaction – reversible vs irreversible reactions – 1 or 2 arrows
24. Acid Base Dissociation Reactions
25. Molar Mass – Atomic mass, Molecular Weight and Formula Weight Calculations
26. Mass Percent and Percent Composition Equation
27. Grams to Moles Conversion and Moles to Gram
28. Mole to Mole and Gram to Gram Stoichiometry
29. Actual Yield, Theoretical Yield, Percent Yield and Percent Error Calculations
30. How To Calculate The Amount of Excess Reactant That Remains
31. How To Identify the Limiting and Excess Reactant
32. Empirical Formula and Molecular Formula Determination Given Grams or Percent Composition
33. Empirical Formula – Combusion Analysis
34. Solubility Rules – Soluble vs Insoluble – Aqueous vs Solid Phase
35. Strong, Weak and Nonelectrolytes – Electrical Conductivity of Solutions
36. Concentration and Molarity Calculations – Moles of Solute and Liters of Solution
37. Dilution Examples Problems – M1V1 M2V2 – Molarity, Volume and Mass in grams
38. Solution Stoichiometry – Limiting and Excess Reactant – Theoretical Yield Calculations
39. Acid Base Titration Problems – M1V1=M2V2
40. Chemical Reactions – Synthesis, Combination, Decomposition, Combustion, Redox Reactions, Single Replacement, Double Replacement Reactions, Acid Base Neutralization, Precipitation Reactions, and Gas Evolution Reactions
41. Net Ionic Equations – Total Ionic and Molecular Equations – Spectator Ions
42. Combined Gas Law Formula and Ideal Gas Law Equation – PV=nRT
43. Boyle’s Law, Charles Law, Gay Lussac’s Law and Avogadro’s Law
44. Pressure, Temperature, Volume and Moles – Direct vs Inverse Relationship
45. Gas Density and Molar Mass Example Problems
46. Dalton’s Law of Partial Pressures and Graham’s Law of Effusion
47. Average Kinetic Energy of a Gas vs Temperature
48. Partial Pressure, Mole Fraction, and Vapor Pressure
49. Gas Law Stoichiometry Problems – STP – Standard Temperature and Pressure
50. Molar Volume – 1 mole of gas = 22.4 Liters
51. Kinetic Molecular Theory of Gases Postulate
52. Units of Pressure – Torr, mm Hg, atm, Kpa, and Pa.
50
views
Soluble and Insoluble Compounds Chart - Solubility Rules Table - List of Salts & Substances
This chemistry video tutorial focuses the difference between soluble and insoluble compounds. It contains a table or chart of the solubility rules and it provides a list of salts and substances - some of which are soluble while others are insoluble. This video contains a worksheet of examples and problems toward the end of the video including answers and solutions.
Useful Textbooks: https://www.amazon.com/shop/theorgani...
E-Book & E-mail Newsletter: https://www.video-tutor.net/math-and-...
Subscribe To My YouTube Channel: https://bit.ly/37WGgXl
_______________________________________________________________________________________________
Here is a list of topics:
1. Understanding The Solubility Rules Table
2. Ions that are always soluble - Na+, K+, Li+, NH4+, C2H3O2-, Cs+, Rb+, ClO4-, ClO3-, and HCO3-
3. Ions that are generally soluble - Cl-, Br-, I- (halides) - Exceptions - Pb2+, Ag+, Hg2 2+
4. Sulfates are generally soluble except with Ba2+, Ca+2, and Sr2+
5. The difference between soluble and insoluble compounds - aqueous vs solid phases
6. Substances that are generally insoluble - Hydroxides, carbonates, sulfides, and phosphates
________________________________________________________________________________________________
Chemistry - Basic Introduction:
• Chemistry
Protons, Neutrons, & Electrons:
• How To Calculate The Number of Proton...
Writing Formulas - Ionic Compounds:
• Writing Chemical Formulas For Ionic C...
Balancing Chemical Equations:
• How To Balance Chemical Equations
Basic Stoichiometry:
• Stoichiometry Basic Introduction, Mol...
Stoichiometry Practice Problems:
• How To Solve Stoichiometry Problems
Solute, Solvent, & Solution:
• Solute, Solvent, & Solution - Solubi...
Strong & Weak Electrolytes:
• Identifying Strong Electrolytes, Weak...
Molarity Practice Problems:
• Molarity Practice Problems
Ion Concentration In Solutions:
• Ion Concentration in Solutions From M...
Dilution Problems:
• Dilution Problems, Chemistry, Molarit...
Types of Chemical Reactions:
• Types of Chemical Reactions
Solubility Rules:
• Solubility Rules
Predicting The Products of Reactions:
• Predicting The Products of Chemical R...
Activity Series of Metals:
• Activity Series of Metals - Chemistry
Will This Reaction Occur?
• Chemistry - Will The Reaction Occur?
Predicting Products of SR Reactions:
• Predicting Products of Single Replace...
Double Replacement Reactions:
• Introduction to Double Replacement Re...
Net Ionic Equations:
• Precipitation Reactions & Net Ionic E...
Writing Chemical Equations From Words:
• How To Write Chemical Equations From ...
Na2CO3 + HCl:
• Na2CO3 + HCl - Sodium Carbonate + Hyd...
Solution Stoichiometry:
• Solution Stoichiometry - Finding Mol...
Molarity & Dilution Problems:
• Molarity Dilution Problems Solution S...
Acid Base Neutralization Reactions:
• Acid Base Neutralization Reactions & ...
Acid Base Titration Problems:
• Acid Base Titration Problems, Basic I...
Mixture Problems:
• Mixture Problems
Calculating Oxidation Numbers:
• How To Calculate Oxidation Numbers - ...
Oxidation and Reduction Reactions:
• Oxidation and Reduction Reactions - B...
Balancing Redox Reactions:
• Half Reaction Method, Balancing Redox...
Ideal Gas Law Problems:
• Ideal Gas Law Practice Problems
General Chemistry 1 Final Exam Review:
• General Chemistry 1 Review Study Guid...
Affiliate Marketing Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
44
views
Graham's Law of Effusion Practice Problems, Examples, and Formula
This graham's law of effusion chemistry video tutorial contains the plenty of examples and practice problems for you to work. It contains the equation or formula for graham's law of effusion in addition to discussion the fundamentals and concepts that are associated with the rate of effusion of a gas from one compartment into another compared to the molar mass of a gas and time.
Useful Textbooks: https://www.amazon.com/shop/theorgani...
E-Book & E-mail Newsletter: https://www.video-tutor.net/math-and-...
Subscribe To My YouTube Channel: https://bit.ly/37WGgXl
________________________________________________________________________________________________
Here is a list of topics:
1. How To Calculate the Rate of Effusion of Helium Gas Given Argon
2. The relationship Between the Rate of Effusion and Molar Mass
3. Effusion vs Diffusion Definition, Concept, and Examples
4. How To Identify The Unknown Gas Using Graham's Law of Effusion By Calculating Molar Mass
5. How to find the time it takes for a gas to effuse from compartment into another.
________________________________________________________________________________________________
Chemistry - Basic Introduction:
• Chemistry
Balancing Chemical Equations:
• How To Balance Chemical Equations
Basic Stoichiometry:
• Stoichiometry Basic Introduction, Mol...
Stoichiometry Practice Problems:
• How To Solve Stoichiometry Problems
Molarity Practice Problems:
• Molarity Practice Problems
Net Ionic Equations:
• Precipitation Reactions & Net Ionic E...
______________________________________________________________________________________________
Pressure & Boiling Point:
• Introduction to Pressure - Force & Ar...
Gas Pressure Unit Conversion:
• Gas Pressure Unit Conversions - torr ...
Manometers & Barometers:
• Manometer Pressure Problems, Introduc...
Water Height & Mercury Column:
• Height of Water in a Barometer Given ...
Boyle's Law Practice Problems:
• Boyle's Law Practice Problems
How Does a Bike Pump Work?
• How Does a Bike Pump Work?
Charles Law:
• Charles' Law
Gay Lussac's Law:
• Gay Lussac's Law Practice Problems
Avogadro's Law:
• Avogadro's law Practice Problems
Ideal Gas Law Problems:
• Ideal Gas Law Practice Problems
Combined Gas Law Problems:
• Combined Gas Law Problems
Gas Stoichiometry Problems:
• Gas Stoichiometry Problems
Molar Mass of a Gas at STP:
• Molar Mass of a Gas at STP - Equation...
Gas Density at STP:
• Gas Density and Molar Mass Problems a...
Dalton's Law of Partial Pressure:
• Dalton's Law of Partial Pressure Prob...
Collecting Gas Over Water:
• Collecting Gas Over Water Practice Pr...
Gas Density of Mixtures:
• Gas Density & Average Molar Mass of a...
Average Kinetic Energy of a Gas:
• Average Kinetic Energy of a Gas and R...
Graham's Law of Effusion:
• Graham's Law of Effusion
Kinetic Molecular Theory of Gases:
• Kinetic Molecular Theory of Gases - P...
Gas Law Problems Review:
• Gas Law Problems Combined & Ideal - D...
___________________________________________________________________________________________
First Law of Thermodynamics:
• First Law of Thermodynamics, Basic In...
Basic Calorimetry Problems:
• How To Solve Basic Calorimetry Proble...
Enthalpy Problems:
• Enthalpy Change of Reaction & Formati...
General Chemistry 1 Final Exam Review:
• General Chemistry 1 Review Study Guid...
____________________________________________________________________________________________
Affiliate Marketing Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
39
views
Gas Density and Molar Mass Formula, Examples, and Practice Problems
This gas density chemistry video tutorial provides the formula and equations for the calculation of the molar mass of a gas and it's density. The gas density formula is derived from the ideal gas law equation. This video contains a worksheet of examples and practice problems for you to work on especially if you need help with mastering chemistry. The answers / solutions are provided in the video tutorial as well.
Useful Textbooks: https://www.amazon.com/shop/theorgani...
E-Book & E-mail Newsletter: https://www.video-tutor.net/math-and-...
Subscribe To My YouTube Channel: https://bit.ly/37WGgXl
________________________________________________________________________________________________
Here is a list of topics:
1. How To Calculate Gas Density of Nitrogen gas at STP Using Dimensional Analysis - 1 mole of gas = 22.4 Liters - Molar Volume
2. Gas Density Formula using Ideal Gas Law Equation PV=nRT
3. Units of density - g/L
4. How To Find The Molar Mass of a Gas Given The Mass in Grams
5. Calculating Molar Mass Using Gas Density at STP
6. How To Determine The Molar Mass of a Gas Given Density at Non standard temperature and pressure conditions
________________________________________________________________________________________________
Chemistry - Basic Introduction:
• Chemistry
Balancing Chemical Equations:
• How To Balance Chemical Equations
Stoichiometry Practice Problems:
• How To Solve Stoichiometry Problems
Molarity Practice Problems:
• Molarity Practice Problems
Net Ionic Equations:
• Precipitation Reactions & Net Ionic E...
______________________________________________________________________________________________
Pressure & Boiling Point:
• Introduction to Pressure - Force & Ar...
Gas Pressure Unit Conversion:
• Gas Pressure Unit Conversions - torr ...
Manometers & Barometers:
• Manometer Pressure Problems, Introduc...
Water Height & Mercury Column:
• Height of Water in a Barometer Given ...
Boyle's Law Practice Problems:
• Boyle's Law Practice Problems
How Does a Bike Pump Work?
• How Does a Bike Pump Work?
Charles Law:
• Charles' Law
Gay Lussac's Law:
• Gay Lussac's Law Practice Problems
Avogadro's Law:
• Avogadro's law Practice Problems
Ideal Gas Law Problems:
• Ideal Gas Law Practice Problems
Combined Gas Law Problems:
• Combined Gas Law Problems
Gas Stoichiometry Problems:
• Gas Stoichiometry Problems
Molar Mass of a Gas at STP:
• Molar Mass of a Gas at STP - Equation...
Gas Density at STP:
• Gas Density and Molar Mass Problems a...
Dalton's Law of Partial Pressure:
• Dalton's Law of Partial Pressure Prob...
Collecting Gas Over Water:
• Collecting Gas Over Water Practice Pr...
Gas Density of Mixtures:
• Gas Density & Average Molar Mass of a...
Average Kinetic Energy of a Gas:
• Average Kinetic Energy of a Gas and R...
Graham's Law of Effusion:
• Graham's Law of Effusion
Kinetic Molecular Theory of Gases:
• Kinetic Molecular Theory of Gases - P...
Gas Law Problems Review:
• Gas Law Problems Combined & Ideal - D...
___________________________________________________________________________________________
First Law of Thermodynamics:
• First Law of Thermodynamics, Basic In...
Basic Calorimetry Problems:
• How To Solve Basic Calorimetry Proble...
Specific Heat Capacity:
• Specific Heat Capacity Problems & Cal...
General Chemistry 1 Final Exam Review:
• General Chemistry 1 Review Study Guid...
____________________________________________________________________________________________
Affiliate Marketing Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
52
views
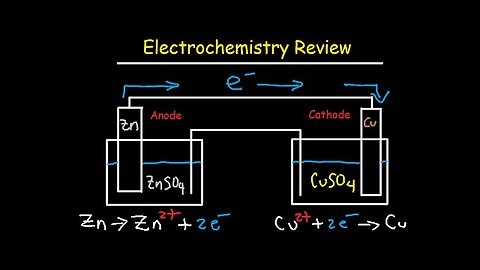
Electrochemistry Review - Cell Potential & Notation, Redox Half Reactions, Nernst Equation
This electrochemistry review video tutorial provides a lot of notes, equations, and formulas that you need to pass your next chemistry test / exam. It’s a nice video that provides an introduction or overview of electrochemistry including the most important fundamental topics and concepts. It contains plenty of examples and practice problems.
Chemistry PDF Worksheets: https://www.video-tutor.net/chemistry...
Here is a list of topics:
1. How To Calculate The Standard Cell Potential
2. Voltaic and Galvanic Cells vs Electrolytic Cells
3. Redox Half Reactions – Oxidation vs Reduction – Anode & Cathode Electrodes
4. Calculating Gibbs Free Energy Using The Cell Potential – delta G & Electrical Work
5. How To Find The Equilbrium Constant K Using Cell Potential and Delta G
6. Nernst Equation – Calculating Nonstandard Cell Potential Using Reaction Quotient Q
7. Calculating The Product Reactant Ratio Using The Cell Potential
8. The Purpose of Salt Bridge – Cations and Anions – Ionic Flow
9. How To Write The Standard Cell Notation For a Galvanic Cell
10. How To Balance Redox Reactions In Acidic Solution and Basic Solution
11. Electrolysis Problems – How To Find The Reactions That Occurs at The Anode and Cathode
12. How To Identify The Strongest Oxidizing Agent and Reducing Agent
13. How To Calculate The Oxidation State / Number
14. Spontaneity – Spontaneous and Nonspontaneous Redox Reactions
15. Equilibrium, Le Chatelier’s Principle and Cell Potential
16. The effect of Reactant and Product Concentration on The Cell Potential
17. Electrochemistry Stoichiometry Problems
18. How To Calculate the Current in Amps Given the mass in grams and the time in minutes
19. Q=It, Charge = Current x time – Faraday’s constant – 96485 Coulombs per mole of electrons
20. Mass in grams, Charge in Coulombs, Current in Amps
Intro to Galvanic & Voltaic Cells:
• Introduction to Galvanic Cells & Volt...
Standard Reduction Potentials:
• Standard Reduction Potentials of Half...
Cell Potential Problems:
• Cell Potential Problems - Electrochem...
Cell Notation Problems:
• Cell Notation Practice Problems, Volt...
Concentration Cells:
• Concentration Cells & Cell Potential ...
Cell Potential & Gibbs Free Energy:
• Cell Potential & Gibbs Free Energy, S...
Cell Potential & Equilibrium K:
• Equilibrium Constant K & Cell Potenti...
Nernst Equation:
• Nernst Equation Explained, Electroche...
Electrolysis of Water:
• Electrolysis of Water - Electrochemistry
Intro to Electroplating:
• Introduction to Electroplating - Elec...
Electrolysis & Electroplating Problems:
• Electrolysis & Electroplating Practic...
Electrochemistry Practice Problems:
• Electrochemistry Practice Problems - ...
______________________________________________________________________________________________
Chemistry - Basic Introduction:
• Chemistry
Molarity, Molality, Density, & Mass %:
• Molarity, Molality, Volume & Mass Per...
Colligative Properties:
• Colligative Properties - Boiling Poin...
General Chemistry 1 Final Exam Review:
• General Chemistry 1 Review Study Guid...
_______________________________________________________________________________________________
Chemical Kinetics - Initial Rate Method:
• Chemical Kinetics - Initial Rates Method
Chemical Equilibrium & Ice Tables:
• Chemical Equilibrium Constant K - Ice...
Acids and Bases - Practice Test:
• Acids and Bases Review - General Chem...
Ksp - Molar Solubility & Ice Tables:
• Ksp - Molar Solubility, Ice Tables, &...
Gibbs Free Energy, Entropy & Enthalpy:
• Gibbs Free Energy - Entropy, Enthalpy...
Entropy - 2nd Law of Thermodynamics:
• Entropy - 2nd Law of Thermodynamics -...
General Chemistry 2 Final Exam Review:
• General Chemistry 2 Review Study Guid...
______________________________________________________________________________________________
SAT Chemistry Subject Test Review:
• SAT Chemistry Subject Test
Coordinate Covalent Bond:
• What is a Coordinate Covalent Bond?
Beer Lambert's Law:
• Beer Lambert's Law, Absorbance & Tran...
_______________________________________________________________________________________________
ACT Math Practice Test:
• ACT Math Test Prep
79
views
How To Balance Redox Equations In Basic Solution
This chemistry video tutorial shows you how to balance redox reactions in basic solution. The first step is to separate the net reaction into two separate half reactions - Oxidation and Reduction. Balance the atoms first under acidic conditions using H+ and H2O and then balance the charges by adding electrons to the side of the chemical equation with the highest oxidation state. Once the electrons in both half-reactions are equal, the two reactions may be combined together to form the net reaction. Add OH- ions to both sides of the equation to neutralize the acid - this is how you can balance the redox reaction under basic conditions.
55
views
1
comment
Oxidizing Agents and Reducing Agents
This video tutorial shows you how to identify the oxidizing and reducing agent in a redox reaction. It has plenty of examples and practice problems for you to work. The first step in determining which species is the oxidizing agent is to start by finding which reactant was reduced. Likewise, the molecule or element that is was oxidized is also the reducing agent. That's how you can determine it. However, you need to calculate the oxidation numbers or states for every element in the chemical equation. Oxidation involves a loss of electrons and occurs when the oxidation number increases. Reduction involves a gain of electrons and occurs when the oxidation state decreases.
Useful Textbooks: https://www.amazon.com/shop/theorgani...
E-Book & E-mail Newsletter: https://www.video-tutor.net/math-and-...
Subscribe To My YouTube Channel: https://bit.ly/37WGgXl
________________________________________________________________________________________________
Chemistry - Basic Introduction:
• Chemistry
Protons, Neutrons, & Electrons:
• How To Calculate The Number of Proton...
Writing Formulas - Ionic Compounds:
• Writing Chemical Formulas For Ionic C...
Balancing Chemical Equations:
• How To Balance Chemical Equations
Basic Stoichiometry:
• Stoichiometry Basic Introduction, Mol...
Stoichiometry Practice Problems:
• How To Solve Stoichiometry Problems
Solute, Solvent, & Solution:
• Solute, Solvent, & Solution - Solubi...
Strong & Weak Electrolytes:
• Identifying Strong Electrolytes, Weak...
Molarity Practice Problems:
• Molarity Practice Problems
Ion Concentration In Solutions:
• Ion Concentration in Solutions From M...
Dilution Problems:
• Dilution Problems, Chemistry, Molarit...
Types of Chemical Reactions:
• Types of Chemical Reactions
Solubility Rules:
• Solubility Rules
Predicting The Products of Reactions:
• Predicting The Products of Chemical R...
Activity Series of Metals:
• Activity Series of Metals - Chemistry
Will This Reaction Occur?
• Chemistry - Will The Reaction Occur?
Predicting Products of SR Reactions:
• Predicting Products of Single Replace...
Double Replacement Reactions:
• Introduction to Double Replacement Re...
Net Ionic Equations:
• Precipitation Reactions & Net Ionic E...
Writing Chemical Equations From Words:
• How To Write Chemical Equations From ...
Na2CO3 + HCl:
• Na2CO3 + HCl - Sodium Carbonate + Hyd...
Solution Stoichiometry:
• Solution Stoichiometry - Finding Mol...
Molarity & Dilution Problems:
• Molarity Dilution Problems Solution S...
Acid Base Neutralization Reactions:
• Acid Base Neutralization Reactions & ...
Acid Base Titration Problems:
• Acid Base Titration Problems, Basic I...
Mixture Problems:
• Mixture Problems
Calculating Oxidation Numbers:
• How To Calculate Oxidation Numbers - ...
Oxidation and Reduction Reactions:
• Oxidation and Reduction Reactions - B...
Balancing Redox Reactions:
• Half Reaction Method, Balancing Redox...
Ideal Gas Law Problems:
• Ideal Gas Law Practice Problems
General Chemistry 1 Final Exam Review:
• General Chemistry 1 Review Study Guid...
Affiliate Marketing Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
88
views
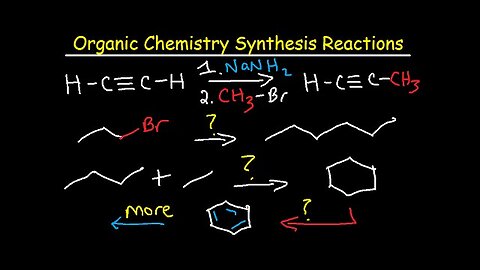
Organic Chemistry Synthesis Reactions - Examples and Practice Problems - Retrosynthesis
This organic chemistry video tutorial focuses on multistep synthesis reactions and retrosynthesis problems. It contains plenty of tips, techniques, examples and practice problems for you to work on.
Here is a list of topics included in this video:
1. Alkyne Synthesis Reactions Using Acetylene
2. Alkynes into Cis and Trans Alkenes
3. Retrosynthesis Reactions With Grignard Reagents
4. Alcohol Synthesis Using Grignard and Organolithium Reagents
5. Ketones and Aldehydes Into Alcohols
6. Synthesis of Ketones From Esters Using The Gilman Reagent
7. Synthesis of Aldehydes From Acid Chlorides Using DIBAH
8. Coupling Alkyl Halides Together Using Organo Copper lithium Reagents - R2CuLi
9. Formation of the Gilman and Grignard Reagent From an Alkyl Halide
10. Alkene Synthesis Reactions From Alkanes
11. Synthesis of Alcohols and Ethers from Alkanes
12. Oxymercuration Demercuration Reaction
13. Hydroboration Oxidation Reaction
14. Alkenes From Alkyl Halides Using The E2 Elimination Reaction
15. Oxidation of Primary Alcohols Into Aldehydes Using PCC
16. Oxidation of Secondary Alcohols Into Ketones Using H2CrO4 or the Jones Reagent
17. Formation of Cycloalkanes From Alkanes Using The Diels Alder Reaction
18. Reductive Amination: Conversion of Ketones to Imines Followed by Reduction to Primary Amines
19. Oxidation of Primary Alcohols Into Carboxylic Acids Using H2CrO4
20. Conversion of Carboxyl Group into Acid Chlorides Followed By Esters, Amides, and Anhydrides
21. Alkyne Synthesis From Alkenes Using The Halogenation Reaction Followed By E2 Elimination Using NaNH2
22. Formation of Alkenes From Ketones Using The Wittig Reaction
23. Synthesis of Benzene From Cyclohexane Using NBS and NaOH
Organic Chemistry PDF Worksheets: https://www.video-tutor.net/orgo-chem...
Organic Chemistry Exam 1 Playlist: https://bit.ly/3kJnNXU
Organic Chemistry 1 Exam 2 Playlist: https://bit.ly/3PKEApB
List of Extended Organic Chemistry Videos:
• List of Organic Chemistry Videos on P...
________________________________________________________________________________________________
Organic Chemistry - Basic Introduction:
• Organic Chemistry - Basic Introduction
How To Draw Lewis Structures:
• Organic Chemistry - How To Draw Lewis...
Functional Groups Review:
• Functional Groups
How To Calculate Formal Charge:
• How To Calculate The Formal Charge of...
Hybridization of Atomic Orbitals:
• Hybridization of Atomic Orbitals - Si...
Resonance Structures:
• Resonance Structures
Acids and Bases - Introduction:
• Acids and Bases - Basic Introduction ...
pKa Values of Acids:
• pKa Values of Acids - Organic Chemistry
Constitutional Isomers:
• Constitutional Isomers
IUPAC Nomenclature of Alkanes:
• IUPAC Nomenclature of Alkanes - Namin...
Newman Projections:
• Newman Projections
Chair Conformations:
• Chair Conformation and Ring Flips
Organic Chemistry Exam 1 Review:
• Organic Chemistry Exam 1 Review
Organic Chemistry Exam 1 Questions:
• Organic Chemistry Exam 1 - IUPAC Nome...
Stereochemistry R/S Configuration:
• Stereochemistry - R S Configuration &...
Optical Activity & Specific Rotation:
• Optical Activity - Specific Rotation ...
SN1, SN2, E1, E2 Reaction Mechanisms:
• SN2 SN1 E1 E2 Reaction Mechanisms Mad...
SN1, SN2, E1, E2 - Practice Test:
• SN1 SN2 E1 E2 Reaction Mechanism - Te...
Alkene Reactions Review:
• Alkene Reactions
Alkyne Reactions Review:
• Alkyne Reactions
Organic Chemistry Exam 2 Review:
• Organic Chemistry 1 Exam 2 Review
Organic Chemistry Exam 2 Questions:
• Organic Chemistry 1 Exam 2 Review Que...
Alcohol Reactions:
• Alcohol Reactions - HBr, PBr3, SOCl2
Free Radical Reactions:
• Free Radical Reactions
Organic Chemistry Reactions Summary:
• Organic Chemistry Reactions Summary
Organic Chemistry 1 Final Exam Review:
• Organic Chemistry 1 Final Exam Review
Organic Chemistry 2 Final Exam Review:
• Organic Chemistry 2 Final Exam Review
270
views
Theoretical, Actual, Percent Yield & Error - Limiting Reagent and Excess Reactant That Remains
This chemistry video tutorial focuses on actual, theoretical and percent yield calculations. It shows you how to determine the percent error using a formula and how it is related to the percent yield. This video also shows you how to identify the limiting reactant by calculating which reagent has the lowest mole per coefficient ratio. It also shows you how to calculate the amount of excess reactant that actually reacts and how much is left over or remains after the reaction is complete. This video provides a few examples and practice problems for you to work on.
Here is a list of topics:
1. How To Write and Balanced a Combustion Reaction
2. How To Identify The Limiting Reagent and Excess Reactant By Calculating The Mole Per Coefficient Ratio
3. How To Calculate Theoretical Yield Using The Limiting Reactant
4. Grams to Grams Conversion Stoichiometry
5. How To Use The Molar Ratio To Convert From One Substance To Another
6. Understanding The Stoichiometric Quantitative Relationship Between The Reactants and Products
7. How To Calculate Percent Yield Using Actual Yield and Theoretical Yield
8. How To Calculate Percent Error From Percent Yield
9. How To Determine How Much Excess Reactant Reacts
10. How To Calculate The Amount of Excess Reactant That Remains or That is Left Over
11. How To Calculate Actual Yield Given Percent Efficiency
Organic Chemistry Tutor - Playlists: https://www.video-tutor.net/
General Chemistry Tutors: https://bit.ly/3PRCtSh
Useful Textbooks: https://www.amazon.com/shop/theorgani...
Introduction to Moles:
• Introduction to Moles
How To Calculate The Molar Mass:
• How To Calculate The Molar Mass of a ...
How To Convert Grams to Moles:
• How To Convert Grams To Moles - VERY ...
How To Convert Moles to Grams:
• How To Convert Moles to Grams
Moles to Atoms Conversion:
• Moles To Atoms Conversion - Chemistry
Grams to Molecules Conversion:
• Grams to Molecules and Molecules to G...
Grams to Atoms:
• How To Convert Grams to Atoms - THE E...
Moles, Atoms, & Grams Conversions:
• How To Convert Between Moles, Atoms, ...
How To Balance Chemical Equations:
• How To Balance Chemical Equations
Stoichiometry - Basic Introduction:
• Stoichiometry Basic Introduction, Mol...
Avogadro's Number:
• Avogadro's Number, The Mole, Grams, A...
Limiting Reactant Problems:
• Limiting Reactant Practice Problems
Excess Reactant Problems:
• How To Find The Amount of Excess Reac...
Theoretical & Percent Yield:
• How To Calculate Theoretical Yield an...
Percent Error:
• Percent Error Made Easy!
Percent Composition By Mass:
• Percent Composition By Mass
Empirical Formula Problems:
• Empirical Formula & Molecular Formula...
Combustion Analysis:
• Introduction to Combustion Analysis, ...
Stoichiometry Practice Test:
• How To Solve Stoichiometry Problems
GPA Calculator:
• How To Calculate Your GPA In College
Save Money In College:
• How To Save Money and Time as a Colle...
SAT Test Prep:
• Evaluating Functions and Solving Basi...
ACT Test Prep:
• ACT Math Test Prep
Resource Links: ----------------------------------------------------------------------------------------------------------------------------------
General Chemistry Help: https://bit.ly/44YpRg6
Useful Chemistry Notes: https://etsy.me/3PoX0fe
Online Courses: http://bit.ly/3RqVN9Z
Buy Now, Pay Later: https://bit.ly/46gRXVg
Affordable Furniture: https://www.wayfair.com/
Car Insurance Student Discount: https://on.gei.co/3rlkHgz
Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
40
views
1
comment
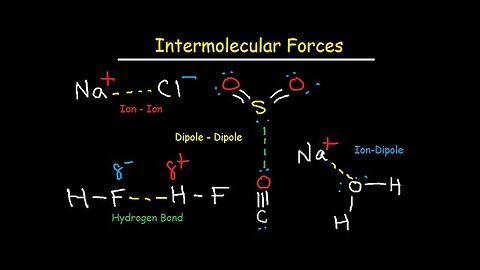
Intermolecular Forces - Hydrogen Bonding, Dipole-Dipole, Ion-Dipole, London Dispersion Interactions
This chemistry video tutorial focuses on intermolecular forces such hydrogen bonding, ion-ion interactions, dipole dipole, ion dipole, london dispersion forces and van deer waal forces. It contains plenty of examples and practice problems to help you understand the most important concepts related to this material.
Chemistry Basic Introduction and Final Exam Review:
https://www.video-tutor.net/chemistry...
My Twitter Page:
https://twitter.com/OrgoChemTutor21
Here is a list of topics:
1. Ion - Ion dipole interactions of KF and CaO
2. Electrostatic Force and Lattice Energy- The effect of charge and ionic radii or size
3. How To Determine Which Ionic Compound has a Higher Melting Point - NaF vs KCl
4. Ion-Dipole Interactions - NaCl and H2O
5. Definition of a Dipole - Polar Molecules & Charge Separation
6. Dipole-Dipole Interactions of Polar Molecules - Partial Charge Electrostatic Attractions of CO
7. Hydrogen Bonding between Hydrogen, Nitrogen, Oxygen, and Fluorine
8. Intermolecular Forces vs Intramolecular Forces
9. Hydrogen Bonding vs Polar & Nonpolar Covalent Bonds
10. London Dispersion Forces & Van Der Waals Forces
11. Permanent Dipoles and Temporary Induced Dipoles - Distribution of electrons in electron cloud
12. Difference Between Atoms and Ions - Cations vs Anions - Number of Electrons and Protons
13. The relationship between Polarizability and Dispersion Forces
14. How To Determine the Strongest Intermolecular Forces In Compounds Such as MgO, KCl, H2O, CH4, CO2, SO2, HF, CH3OH, LiCl, CH2O, CO, and I2
15. The relationship between Boiling Point and Vapor Pressure
16. Straight Chained vs Branched Alkanes - Boiling Point and Intermolecular Forces - Surface Area
17. Ranking Boiling Point In Order of Increasing Strength for I2, Br2, F2, and Cl2
18. Polar and Nonpolar Organic Compounds - Polarity and Water Solubility
19. Ranking Boiling In Decreasing Order For HF, HCl, HBr, and HI
20. The effect of Molar Mass and Number of electrons on the Overall Intermolecular Force / LDF
53
views
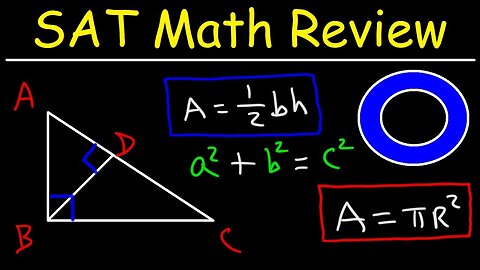
SAT Math Test Prep Online Crash Course Algebra & Geometry Study Guide Review, Functions,Youtube
This online sat math test prep review youtube video tutorial will help you to learn the fundamentals behind the main concepts that are routinely covered on the scholastic aptitude test. This online crash course video contains plenty of examples and practice problems for you work on including very hard / difficult math questions with answers and solutions included. There are six main lessons in this study guide that are accompanied by a review of the most important topics, concepts, equations and formulas that you need to do well on the sat. This video contains plenty of multiple choice problems that you can work on as a practice test.
Full 8 Hour SAT Video on Patreon:
https://bit.ly/3sadcH0
Printable PDF With All of The Practice Problems of The Full 8 Hour Video:
https://bit.ly/3hbd1Vy
SAT Math Patreon Videos:
• SAT Math - Patreon
My Twitter Page:
https://twitter.com/OrgoChemTutor21
Topics include:
1. Evaluating composite functions and algebraic expressions
2. Solving equations and finding the value of x – SAT Math Algebra Review
3. Simplifying Radicals, Exponents, and Factoring Trinomials
4. Solving Equations With Two Variables Using The Substitution and Elimination Method
5. Absolute Value Equations and Multivariable Functions
6. How To Convert Sentences Into Equations to Solve SAT Math Word Problems
7. Word Problems With Averages and Total Sum
8. Consecutive Even / Odd Positive or Negative Integer Word Problem
9. Basketball Team Word Problems – Games won vs Lost
10. Past Present Future Age Word Problem
11. Inclusive vs Exclusive Numbers In a List
12. Distance Rate Time SAT Math Word Problems
13. Ratios and Proportions Based Word Problems
14. Red Green Blue Yellow Marble Probability Problem
15. Nickels Dimes Quarters Word Problem
16. Number of Workers SAT word problem
17. Permutations Combinations and Fundamental Counting Principle
18. Two or Three Digit Integer Word Problem
19. Average Score, Arithmetic Mean, and Weighted Average SAT Math Problem
20. Percent Increase or Decrease Word Problem – Sales Tax vs Discount
21. Original Selling Price and Percent Change Practice Problems
22. Percentage Problems with x and y Variables
23. Slope Calculations, Midpoint and Distance Formula
24. Mean, Median, Mode, and Range
25. Arithmetic and Geometric Series and Sequences Word Problems
26. Linear, Quadratic, and Absolute Value Functions and Graphs
27. SAT Math Geometry Review
28. How to Find the Side Length of a Triangle Given Area and Height
29. How to Calculate the Perimeter of a Triangle Given Hypotenuse
30. How to Determine the Area of a Right Triangle
31. Calculating the Area of a Parallelogram
32. Percent Increase or Decrease of Radius and Height of a Cylinder – Effect on Volume
33. How to Calculate the Perimeter of a Square Given It’s Diagonal
34. Calculating the Altitude of a 45 45 90 degree right triangle
35. Finding the Perimeter of an Equilateral or Equiangular Triangle
36. Calculating the Sum of the Three Remote Exterior Angles of a Triangle
37. Segment Midpoint Geometry Problems
38. Parallel Lines – Corresponding, Alternate Interior, Exterior and Vertical Angles
39. Linear Pairs – Supplementary and Complementary Angles
40. Finding the area of the shaded region of a circle
41. Tangent and Secant Lines on Circles
42. How to Calculate the Arc of a segment of a circle
43. Rectangle Semicircle Word Problem
44. How to Calculate the Perimeter of a Rhombus Given It’s Side Length
45. How to Determine the altitude of a right triangle using the geometric mean and proportions
Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf should you decide to make a purchase through such websites.
79
views
General Chemistry 2 Review Study Guide - IB, AP, & College Chem Final Exam
This general chemistry 2 final exam ap, ib, & college chem review video tutorial contains many examples and practice problems in the form of a multiple choice practice test. It has about 40 out of 150 test bank problems in the form of a standardized final exam that will help study for your next test. Feel free to use this video as an study guide / online course. It provides the concepts, solutions, as well as the equations and formulas that you need to pass your chemistry class. This video provides a nice overview of Gen Chem 2.
Access The Full 7 Hour Video on Patreon:
https://www.patreon.com/MathScienceTutor
Direct Link to The Full Video:
https://bit.ly/3X0t6AL
PDF Worksheet - 150 Practice Problems:
https://bit.ly/3iuopQz
Here is a list of topics:
1. Chemical Kinetics - Calculating the average rate of appearance and disappearance.
2. How To Find The Rate Law Expressions, Order of Reactants, and the Rate Constant K including units
3. Zero, First, and Second Order Reactions
4. Calculating The Final Concentration of a Zero Order Reaction
5. Calculating The Rate Constant K For a Second Order Reaction Using Half Life
6. How To Calculate The Activation Energy Given The Rate Constant K and Temperature
7. Alpha, Beta, and Gamma Particles. Positron, Protons, Neutrons, and Electrons - How To Identify the Missing Element.
8. First Order Half Life Decay Practice Problems
9. How To Calculate Kp from Kc
10. Hess Law Equilibrium Constant K
11. Calculating Kp from Partial Pressure
12. Chemical Equilibrium and ICE Tables
13. How To Determine If The Reaction Will Shift To The Left or Right Using The Reaction Quotient Q
14. Le Chatelier's Principle - Concentration, Pressure, Volume and Temperature Changes - Effect on the System
15. How To Identify Strong and Weak Acids
16. Acidic, Basic, and Neutral Salts
17. How To Calculate the pH of a weak Acid HC2H3O2, a weak base NaF, and a buffer solution
18. How To Identify a Buffer Solution, Acid Base Titrations
19. How To Identify a Lewis Acid
20. Ksp Equilibrium Expression and Molar Solubility
21. Saturated and Unsaturated Solutions - Solubility Product Constant
22. How To Determine the sign of entropy or delta S from a reaction.
23. Thermodynamics - Gibbs free energy, enthalpy and entropy
24. Spontaneous at low high or at all temperatures
25. How To Calculate delta G from Equilibrium Constant K
26. Spontaneous vs Nonspontaneous reactions
27. Anode vs Cathode - Oxidation vs Reduction
28. How To Balance Redox Reactions In acidic and Basic Conditions
29. delta G and Cell Potential - Faraday's Constant G = -nFE
30. Nonstandard delta G and Cell Potential
31. pH and pOH of solution at the equivalence point
32. pH of solution before, during, and after a titration
33. Weak acid strong base titration vs strong acid weak base titration
34. How To Select Indicator Using pH and pKa
35. Henderson Hasselbach Buffer Equation
36. How To Calculate the Boiling Point Using delta H and S
37. Equilibrium Problems, ICe Table, Quadratic Equations
38. How To Calculate Ksp and Molar Solubility in g/L - common ion effect
39. Le Chatelier's Principle - Acids and Bases, Ksp, delta G and Cell Potential
Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
73
views
General Chemistry 1 Review Study Guide - IB, AP, & College Chem Final Exam
This video tutorial study guide review is for students who are taking their first semester of college general chemistry, IB, or AP Chemistry. Even if you’re studying for the general chemistry section of the MCAT, DAT, PCAT, OAT or SAT Subject chemistry test, this video can help give you a nice overview of all the topics you need to learn in General Chemistry 1. This introduction video contains plenty of examples and practice problems to help prepare you for the final exam. It has about 160 multiple choice questions in the form of a practice test. Feel free to use it as a study guide. The solutions to each problem is provided as well as the equations and formulas that you need to solve it.
Access The Full 8 Hour Video on Patreon:
https://www.patreon.com/MathScienceTutor
Direct Link to The Full Video:
https://bit.ly/3GstGjy
Download The Exam - 160 Practice Problems:
https://bit.ly/3VTpcbj
General Chemistry 2 Review - 7 Hours - 150 Practice Problems:
https://bit.ly/3X0t6AL
Youtube Membership:
/ @theorganicchemistrytutor
General Chemistry 2 Final Exam Review:
• General Chemistry 2 Review Study Guid...
Here is a list of topics:
1. How To Find The Number of Protons, Neutrons, and Electrons In an Atom
2. Nomenclature of Ionic and Molecular Compounds
3. Percent Composition, Stoichiometry, Molarity, Dilution Problems, Acid Base Titrations
4. Oxidation State, Combined Gas Law, Gas Density at STP, Dalton’s Law of Partial Pressure
5. Collected over water gas law problems, How to Identify the Unknown Compound
6. Average Kinetic Energy of Gases, Real Gas vs Ideal Gas High Temperature Low Pressure
7. Specific Heat Capacity, Heat of Fusion, Heat of Combustion, Enthalpy, Products – Reactants,
8. Condensation, Sublimation, Melting, Vaporization, Freezing, Deposition
9. Heat of Formation, Wavelength Frequency and Energy of a photon, Electron Configuration
10. Which four quantum numbers are incorrect? N l ml ms, s p d f orbitals,
11. Electromagnetic radiation – Radio waves Microwaves Infrared Visible Light Ultraviolet X-rays and Gamma rays.
12. Ionization Energy Electronegativity Atomic Radii Ionic Radius Electron Affinity and Metallic Character
13. Hybridization, Molecular Geometry, Lewis Structure, Bond Angle, Polar or Nonpolar
14. SO2, CO2, CH4, BH3, H2O, NH3, BF3, SF6, NF3, SF4, PCl5, XeF4
15. Intermolecular Forces, Hydrogen Bonding, Dipole Dipole Interactions, London Dispersion Forces, Ion Dipole, Permanent Dipoles, Temporary Induce Dipole, Van der Waal Forces, and Ionic Bonds.
16. Highest Boiling Point, Lowest Freezing Point, pH of a solution, Empirical Formula,
17. Molality, Mass Percent, Density, Solutions, Nonelectrolytes, Vapor Pressure, Combustion Analysis
18. Colligative Properties – Boiling Point Elevation, Freezing Point Depression, Osmotic Pressure
19. Average atomic mass and Percent Relative Abundance, Chemical Change, Chemical Formula
20. Actual Yield, Theoretical Yield, and Percent Yield, Oxidizing Agent vs Reducing Agent
21. Grams to moles to molecules to atoms – conversions / dimensional analysis
22. How To Find The Mass of The Excess Reactant That Remains, Limiting Reactants,
23. Homogeneous Mixture – Air plus salt and water, Soluble vs Insoluble
24. Representative Elements, Transition Metals, Alkali Metals, Alkaline Earth Metals, Halogens, Chalcogens, Noble gases, and Inner Transition Metals
25. Redox Reactions, Single Replacement Reactions, Combustion Reactions, Synthesis Reactions, Combination Reactions, and Decomposition Reactions, Double Replacement Reactions
26. Acid Base Neutralization Reactions, Precipitation Reactions, Gas Evolution Reactions
27. How to Identify the Unknown Metal given its Oxide and Formula, Gas Stoichiometry
28. Ideal Gas Law PV=nRT, Molar mass of unknown gas, mole fraction and partial pressure,
29. Root Mean Square Velocity, Graham’s Law – Rate of Effusion, Kinetic Molecular Theory
30. Pressure Volume Temperature Graphs – Boyle’s Law, Charles Law, and Gay Lussac’s Law
31. ionic and covalent bonds, Internal Energy of System,
32. Final Temperature of Water Solution, Hess Law, Thermochemical Equations,
33. Endothermic vs Exothermic, Calorimetry, Lattice Energy, Valence Electrons, Paramagnetic.
34. electron configuration exceptions, noble gas notation
35. Hund’s rule, Heisenberg Uncertainty Principle, Aufbau Principle, Pauli Exclusion Principle
Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
96
views
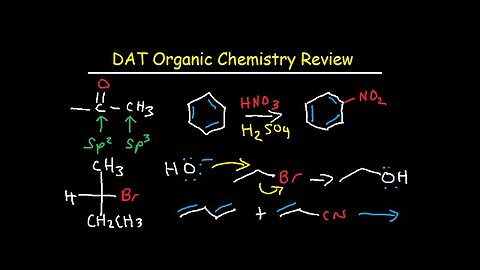
DAT Organic Chemistry Study Guide Review
This DAT prep video tutorial provides a review of organic chemistry for those taking the dental admissions exam. It provides plenty of notes, examples, and practice questions with reaction mechanisms to help you learn the most important topics in organic chemistry. You should definitely incorporate flashcards to help you memorize some important reactions.
Full DAT Course Review:
https://vimeo.com/ondemand/datreviewc...
Here is a list of topics:
1. Hybridization – s, sp, sp2, sp3, Molecular Geometry, and Bond Angle
2. Functional Groups – Ketones, Aldehydes, Ethers, Esters, Nitriles & Amides
3. Formal Charge, Resonance Structures, Boiling Point, & Water Solubility
4. Carbocation, Carbanion, Radical, and Alkene Stability
5. Primary, Secondary, and Tertiary Hydrogen & Carbon atoms, Amines, & Alcohols
6. Acidity & Pka – Brownsted Lowry & Lewis Acids & Bases – Acid Strength
7. Newman Projections, Chair Conformations, Cycloalkanes & Fischer Projections
8. Radical Bromination and Monochlorination Reactions
9. Chiral Carbon Atoms, Stereocenters, Stereoisomers, R & S Absolute Configuration
10. Stereochemistry – Enantiomers, Meso Compounds, Diastereomers and Constitutional Isomers
11. Optical Purity, Enantiomeric Excess, & Observed Rotation
12. Electrophilic Addition Reactions of Alkenes – Hydration & Hydrohalogenation Mechanism
13. Hydroboration Oxidation and Oxymercuration Demercuration Reaction of Alkenes
14. Syn Hydroxylation, Ozonolysis, Carbene Reactions, Hydrogenation, & Epoxidation
15. Hydrogenation of Alkynes Into Alkanes and Cis / Trans Alkenes - Lindlar’s Catalysts vs Na & NH3
16. Alkyne Synthesis Reactions Using Acetylene and NaNH2
17. SN2, SN1, E1, E2 Reaction Mechanism Chart – Polar Protic vs Aprotic Solvents
18. Leaving Groups, Nucleophile Strength, Steric Hindrance, Bulky Bases
19. SN2 Inversion of Stereochemistry vs SN1 Racemic Mixture & Anti E2 Elimination
20. Zaitsev vs Hoffman Product – E2 Reaction
21. Reactions of Alcohols – HBr, PBr3, SOCl2, H2SO4, H3PO4, PCC, H2CrO4, KMnO4, Na2Cr2O7
22. Acid Catalyzed E1 & E2 Dehydration Reactions of Alcohols
23. Hydride Shift, Methy Shift, and Ring Expansion Carbocation Rearrangements
24. Williamson Ether Synthesis Reactions and Bimolecular Dehydration Reaction of Alcohols
25. Acid Catalyzed Cleavage of Ethers With HBr and HI
26. Acid and Base Catalyzed Cleavage of Epoxides With CH3O- and CH3OH / H+
27. Oxidation of Primary and Secondary Alcohols
28. Reduction of Aldehydes & Ketones With NaBH4, LiAlH4,
29. Grignard, Gilman, DiBAH, and Organic Lithium Reagents – Synthesis Practice Questions Included
30. HIO4 Periodic Acid Cleavage of 1,2 Diols
31. 1,3-Butadiene Reactions With Br2, Cl2, and HBr – 1,2 vs 1,4-Addition – Kinetic vs Thermodynamic Products.
32. Diels Alder Reaction – Diene vs Dienophile – Endo vs Exo Product – S-Trans vs S-Cis Conformation
33. UV/Vis Spectroscopy – Conjugated Alkenes, 200-400nm Wavelength
34. Aromatic, Antiaromatic, vs Nonaromatic Compounds – Huckel’s Rule – 4n+2
35. Nomenclature – Benzene Derivatives – Aniline, Anisole, Toluene, Xylene, Styrene, Napthalene
36. Electrophilic and Nucleophilic Aromatic Substitution Reaction Mechanism
37. Nitration, Sulfonation, Friedel Crafts Alkylation & Acylation, Bromination & Chlorination
38. Benzene Reactions – HNO3, H2SO4, CH3Cl / AlCl3, Br2 / FeBr3, SO3 / H2SO4
39. Arenediazonium Salts – NaNO2 / HCl or HNO2 – Sandmeyer Reaction – CuCl, CuBr, CuCN
40. Enolate Ions, LDA, Kinetic vs Thermodynamic Product
41. Alpha Hydrogen Substitution Reactions – Br2, OH-,
42. Acetal Formation, Imine, Enamine, Wittig Reaction, Stork Reaction, and Enol Keto Tautomers
43. Reactions of Amines, Gabriel Synthesis, Nitrile Reduction, Reductive Amination
44. Hoffman and Curtius Rearrangement, Cope & Hoffman Elimination & HVZ Reaction
45. Carboxylic Acid Derivatives – Acid Chlorides, Esters, Anhydrides, and Amides
46. Aldol Reaction, Claisen Condensation, Acetoacetic and Malonic Ester Synthesis Reaction
47. Michael Addition, Intramolecular Aldol, Robinson Annulation, and Dieckmann Reaction
48. H-NMR, C-NMR, MS, and IR Spectroscopy
49. How To Determine The Number of Signals In a H-NMR Spectrum
50. Tollen’s Test, Iodoform Test, 2,4-DNP, Bromine Water, Chromic Acid and Lucas Reagent Test
79
views
Math Videos: How To Learn Basic Arithmetic Fast - Online Tutorial Lessons
This online math video tutorial /lecture shows you how to learn basic arithmetic fast and easy. It contains plenty of examples and practice problems including drill quizzes to help you boost your math skills to the next level. Whether you're high school, college or an adult who wants to learn math, this video is for you.
Learn Useful Study Skills: https://amzn.to/2UTLsbR
Video Playlists: https://www.video-tutor.net
Homework Help: https://bit.ly/Find-A-Tutor
Subscribe: https://bit.ly/37WGgXl
Support & Donations: https://www.patreon.com/MathScienceTutor
Youtube Membership:
/ @theorganicchemistrytutor
Ten Side Hustles For College Students
https://www.video-tutor.net/side-hust...
Here is a list topics:
1. How To Add and Subtract Real Numbers Using a Number Line
2. How To Add Negative Numbers
3. How To Perform Simple Addition and Subtraction By Hand
4. How To Multiply Two Numbers In Your Head – Mental Math
5. Basic Arithmetic Operations – Multiplication and Long Division
6. How To Add, Subtract, Multiply and Divide Fractions
7. How To Convert Improper Fractions Into Mixed Numbers
8. How To Subtract By Borrowing a 1 Method
9. Division of Fractions – Keep Change Flip Principle
10. How To Convert Fractions To Decimals Mentally and Using Long Division
11. How To Divide Two Numbers Using Reverse Multiplication
12. How To Convert Percent Into Decimal
13. How To Find The Percentage of a Number
14. How To Calculate Original and Final Price Using Sales Tax
15. How To Do Simple Multiplication By Using Money as an Illustration
16. Order of Operations Review – PEMDAS
17. How To Simplify Negative and Zero Exponents
18. How To Evaluate Numbers With Fractional Exponents
19. How To Evaluate Logarithms Especially Logs With Fractions
20. How To Find Square Root of a Number
21. Square of Negative Numbers – Imaginary Number i
22. How To Simplify Radical Expressions – Square & Cube Roots Using Perfect Squares & Cubes
23. How To Add and Subtract Radical Expressions
24. How To Convert Scientific Notation to Standard or Decimal
25. How To Multiply and Divide Numbers In Scientific Notation
26. How To Square Root of a Number In Scientific Notation
27. How To Factor Trinomials By Grouping
28. GCF – Greatest Common Factor
29. How To Solve Equations By Factoring – Difference of Perfect Squares Method
30. How To use The Quadratic Formula To Solve Equations
31. How To Solve Linear Equations Containing Decimals, Fractions, and Parentheses
32. How To Graph Linear and Quadratic Equations
33. How To Write The Equation of Line in Point Slope Form and Slope Intercept Form
34. How To Calculate The Slope of a Line Given Two Points
35. How To Graph Parabolic, Quadratic, Cubic, Absolute Value, Rational, and Radical Equations Using Transformations
36. Domain and Range For Quadratic Functions
37. How To Identify The Minimum and Maximum Point of a Quadratic Equation
38. How To Identify The Vertex and Axis of Symmetry of a Quadratic Equation
39. Reflection about the x axis, y axis, and the origin
40. Horizontal and vertical shifts.
41. Parent Functions of Exponential and Logarithmic Graphs and Equations
Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
150
views
H NMR Spectroscopy Review - Examples & Multiple Choice Practice Problems
This organic chemistry video provides a review of H NMR spectroscopy. It provides plenty of examples and multiple choice practice problems that you might encounter on your next test or exam. It discusses chemical shifts for common functional groups such as aldehydes, carboxylic acids, ethers, alkyl halides, and aromatic benzene rings. It provides a review of the H NMR signals and the spin spin splitting pattern of the n+1 rule that you need to know. It also shows you how to determine the number of signals in an h nmr spectrum and discusses the terms upfield and downfield so you can understand it.
My Website: https://www.video-tutor.net
Patreon Donations: https://www.patreon.com/MathScienceTutor
Amazon Store: https://www.amazon.com/shop/theorgani...
Here is a list of topics:
1. Common Chemical Shifts / H NMR Table Values
2. How To Find The Number of Signals
3. N + 1 rule - Spin Spin Splitting
4. How To Identify Identical Hydrogen Atoms
5. How To Draw a HMR Signals /Spectrum
6. Upfield vs Downfield
7. H-NMR Analysis and Integration
Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links.
34
views
Amine Synthesis Reactions Organic Chemistry - Summary & Practice Problems
This organic chemistry video tutorial on amine synthesis reactions covers a variety of topics and includes plenty of examples and practice problems to work on.
Here is a list of topics contained in this video:
1. Nomenclature of Amines - IUPAC & Common Names such as Ethanamine & Ethyl Amine
2. Aromatic Heterocycles - Pyrrole, Pyrrolidine, Pyridine, Piperidine, Pyrimidine, Imidazole, & Purine
3. Physical Properties of Amines - Boiling Point, Water Solubility, Polarity, Hydrogen Bonding
4. Basicity of Amines in Gas Phase & Aqueous Phase
5. Acidity of Amine Conjugate Acid - PKa - Electron Donating / Releasing & Withdrawing Groups
6. Quarternary Ammonium Salts - Phase Transfer Catalysts
7. Synthesis of Amines From Alkyl Halides Using NaCN, NaN3, NH3, and NaNH3 Reaction Mechanism
8. Reduction of Nitriles to Amines Using H2 With Pd, Pt, or Ni Catalyst
9. Reduction of Azides Using LiAlH4 Followed By H2O
10. Gabriel Synthesis Reactions of Primary Amines From Alkyl Halides Using H2N-NH2 or N2H4
11. Synthesis of Amino Acids From Alpha Brominated Carboxylic Acids
12. Hoffman Elimination Reaction - Amines to Alkenes Using Excess CH3I, Ag2O, H2O and Heat
13. Oxidation of Tertiary Amines to Amine Oxides Followed By Cope Elimination
14. Anti vs Syn Elimination Stereochemistry - Cope vs Hoffman - Anti-Zaitsev Regiochemistry
15. Hoffman Rearrangement of Amides to Amines Using Br2 / Cl2, OH-, and H2O
16. Curtius Rearrangement of Acid Chlorides to Amines Using NaN3, H2O, and Heat
17. Formation of Alkynes From Alkyl Halides Using NaNH2
18. Ketones to Imines & Enamines
19. Reductive Amination of Ketones Into Primary & Secondary Amines Using NaBH3CN - Sodium CyanoBorohydride
20. Conjugate Addition of Ketones & Amines
21. Formation of Oximes From Ketones Using Hydroxyl Amine - H2N-OH
22. Electrophilic Aromatic Substitution Reactions of Pyrrole at C-2 Position
23. EAS Reactions of Aromatic Heterocycles Such as Furan and Thiophene
24. Nucleophilic Aromatic Reactions of Pyridine Using NaNH2, NaOH, NaSH, CH3MgBr, and NaOCH3
25. Diazotization of Amines Using NaNO2 and HCl or HNO2 / HONO
26. Benzene to Aniline Reactions
27. Sandmeyer Reaction - CuBr, CuCl, CuCN, H3PO2, H3O+, and HBF4
28. Arenediazonium Salts and Diazocoupling
29. Formation of N-nitrosoamines from Secondary Amines
30. Synthesis Reactions - Alkane to Amine
31. Amines From Amides, Ketones, Azides, Alkyl Halides, Alcohols, Etc.
32. Side Chain Reactions of Aromatic Hetereocycles.
33. Proposed Synthesis of Piperazine From Ethane
34. Pyrrolidine to Pentane Reaction Mechanism
92
views